1. Capping
What it is: Separation of the tablet’s top or bottom layer during ejection or handling.
Why it happens: Entrapped air, insufficient inter-particle bonding, incorrect compression force, worn punches, or poor die condition.
2. Lamination
What it is: Tablet splits into multiple layers, not limited to the cap.
Why it happens: Rapid decompression, excess fines in formulation, trapped air, die wall friction, or poor granulation.
3. Sticking
What it is: Material adheres to the punch tip, predominantly the upper punch.
Why it happens: Inadequate lubrication, presence of low melting point ingredients, poor punch polishing, rough punch surfaces, or high moisture in the blend.
4. Picking
What it is: Small tablet parts stick to the punch face, often near embossed logos.
Why it happens: Poorly engraved punches or sticky formulation components, particularly common with logoed or deeply embossed tablets.
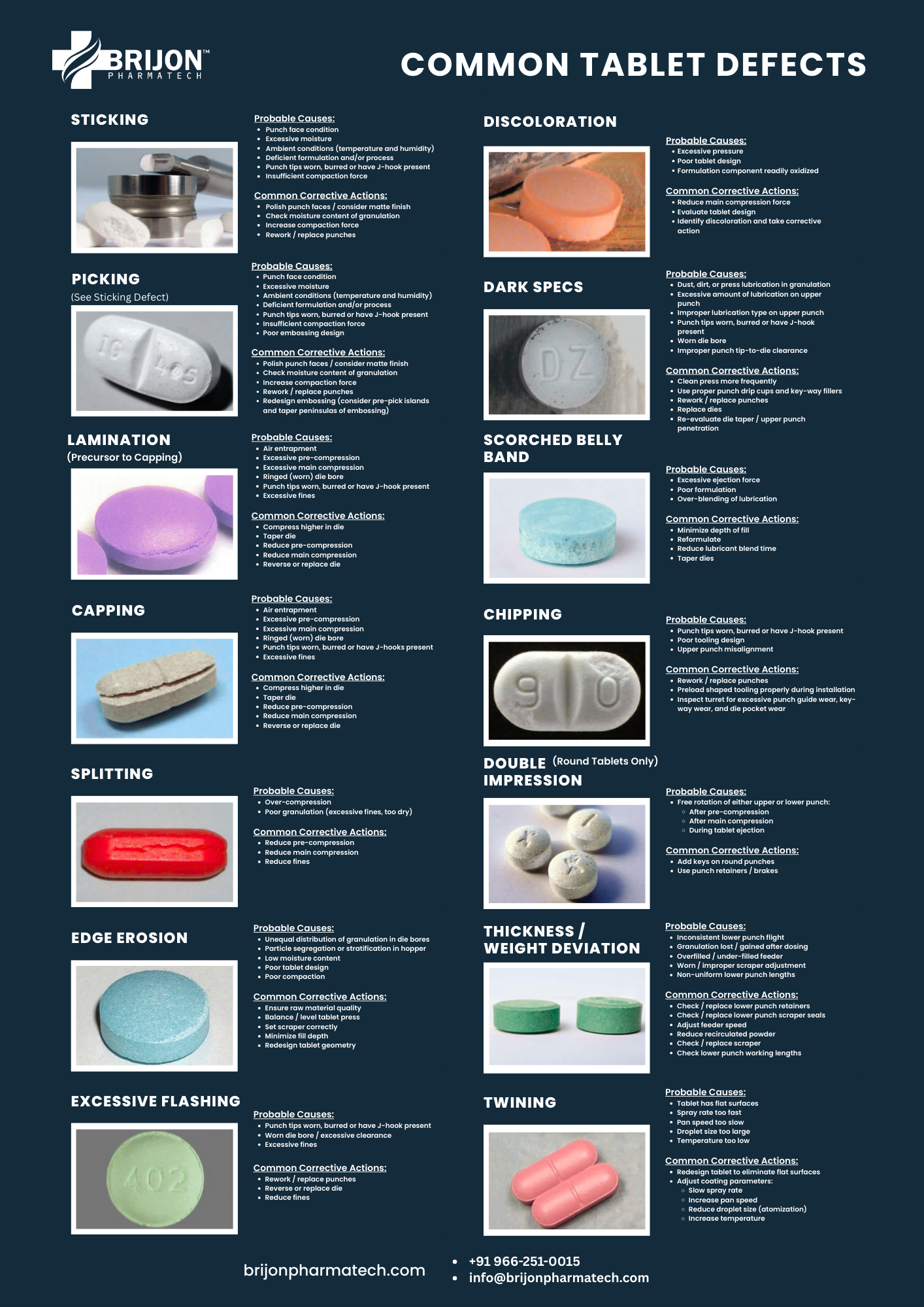
5. Chipping
What it is: Breakage of tablet edges during ejection or handling.
Why it happens: Low binder content, fragile tablet structure, sharp tooling edges, or die misalignment.
6. Cracking
What it is: Visible surface or edge cracks on tablets.
Why it happens: Rapid granule expansion, improper drying, excessive compression, or tooling wear causing uneven pressure distribution.
7. Double Impression
What it is: Duplicate images or logos appear on tablets.
Why it happens: Punch movement during rebound from loose punch fixation or non-keyed tooling setups.
Preventing Defects Starts with Tooling and Formulation Alignment
Addressing tablet defects involves a thorough review of formulation characteristics, machine parameters, and tooling condition. High-quality punches and dies, meticulous polishing, precise alignment, and understanding material behavior during compression are essential measures to minimize defects.
Frequent or widespread manufacturing defects often indicate process inconsistencies rather than material problems alone. Partnering with a reliable tooling supplier and adopting proactive maintenance and process validation can significantly improve tablet quality and production efficiency.
Understanding and preventing tablet defects is integral to delivering quality pharmaceutical products and ensuring patient safety.




